Avelumab Biosimilar - Research Grade
Bulk Avelumab Biosimilar - Research Grade
Product Benefits:ichorbio's Avelumab Biosimilar - Research Grade is manufactured in a cGMP compliant facility. ichorbio's avelumab biosimilar is strictly for research use only (RUO).
Size:ichorbio's research grade avelumab biosimilar is available in the following sizes: 5mg, 10mg, 20mg, 50mg, 100mg ichorbio regularly manufactures bulk multi-gram amounts of our avelumab biosimilar - please contact us for pricing.
Target:PD-L1
Clone:n/a
Isotype:IgG1 lambda
Other Names:Programmed cell death 1 ligand 1, Cd274, PDCD1 ligand 1, Programmed death ligand 1, B7 homolog 1, B7-H1, B7h1, Pdcd1l1, Pdcd1lg1, Pdl1, PDL-1
Host:Humanized
Species Reactivity:Human
Specificity:Detects human PD-L1.
Purification Method:This monoclonal antibody was purified using multi-step affinity chromatography methods such as Protein A or G depending on the species and isotype.
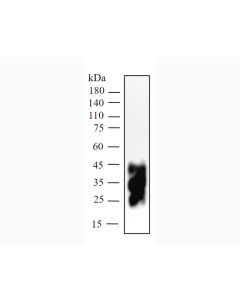
Background:Avelumab biosimilar is a programmed death ligand-1 (PD-L1) blocking antibody. Avelumab is a human IgG1 lambda monoclonal antibody that has a molecular weight of approximately 147 kDa.
Concentration:1.0 - 5.0 mg/ml
Formulation:Sterile, no preservative, no pyrogen, clear, colorless to slightly yellow liquid, which contains d-mannitol, glacial acetic acid, polysorbate 20 and sodium hydroxide, the pH range is 5.0-5.6. BSA and Azide free.
Purity:>95% by SDS-PAGE and HPLC
Endotoxin:≤ 0.75 EU/mg as determined by the LAL method
Storage:Avelumab Biosimilar - Research Grade is stable for at least four (4) weeks when stored sterile at 2-8°C. For long term storage aseptically aliquot in working volumes without diluting and store at –80°C. Avoid Repeated Freeze Thaw Cycles.
Applications:Functional Assays
Application Notes:Each investigator should determine their own optimal working dilution for specific applications.
Use: ichorbio's avelumab biosimilar is for research use only (RUO). Recent Research into Avelumab: Avelumab is a human IgG1 monoclonal antibody directed against the immunosuppressive ligand PD-L1 (programmed death-ligand 1) that prevents its interaction with the inhibitory T-cell receptor, PD-1 (programmed cell death protein 1). Upon binding, avelumab promotes T-cell reactivation and restores T-cell-mediated immune response against tumor cells. Avelumab is also the only PD-L1 antibody so far to also induce antibody-dependent cell-mediated cytotoxicity (ADCC) against tumor cells expressing PD-L1. It was first approved by the FDA in March 2017 for the treatment of patients with metastatic MCC (Merkel cell carcinoma) and later for advanced types of renal cell carcinoma and urothelial cancer. Here are some examples of avelumab’s latest research on advanced and metastatic cancer treatments. In one of the latest studies published in Cell, Chew et al. (2020) hypothesized that inhibition of endocytosis may improve the efficiency of immunotherapeutic antibodies on the clinical outcome of advanced cancer patients. Using PCZ, a dynamin inhibitor in combination with avelumab, they significantly improved the therapeutic response in mice with CT26.WT tumors in terms of tumor growth and clearance. Their data in vivo showed that this was due to an increase in avelumab-mediated ADCC and the induction of a potent immune response against tumor cells. In this study, the temporary inhibition of endocytosis increased tumor cell antigen presentation and target availability for immunotherapeutic agents such as cetuximab, trastuzumab, and avelumab. In early 2020, Tafuri et al. performed a meta-analysis on the response rates of patients treated with PD-1 and PD-L1 inhibitors with urothelial carcinoma refractory to standard treatment. The study focused on five FDA-approved antibodies targeting PD-1 or PD-L1: atezolizumab, durvalumab, avelumab, nivolumab, and pembrolizumab. The authors analyzed objective and complete responses, as well as median overall survival and progression-free survival. They measured higher estimated response rates for the selected antibodies than for the “other salvage therapies”. In January 2020, Elbers et al. published the results of a phase-I feasibility trial for advanced-stage head and neck squamous cell carcinoma (HNSCC) patients unfit for cisplatin treatment. The alternative treatment for these patients consists of radiotherapy with cetuximab but the low 5-year overall survival still highlights the need to improve the current therapy strategy. In this study, the authors tested a combination of cetuximab and PD-L1 immune checkpoint blockade with avelumab. They observed transient and manageable immune-related toxicity that did not impact the toxicity of radiotherapy alone. Based on their results, they concluded that the combination treatment was feasible and needed to be tested on a larger scale. At around the same time, another group conducted a meta-analysis with 6,896 participants in 26 different studies to investigate the incidence and potential factors of fatal adverse events associated with the use of PD-L1 inhibitors for the treatment of patients with advanced cancer. The search was performed on PubMed, Embase, and the Cochrane Library and included four PD-L1 inhibitors: atezolizumab, durvalumab, avelumab, and cemiplimab. The overall incidence of fatal adverse events associated with PD-L1 inhibitors was 1.24%. Interestingly, the incidence of fatal adverse events associated with avelumab was lower than that of both atezolizumab and durvalumab. Keywords: PDL1; PD1; immune checkpoints; avelumab; advanced tumors. References: Akintunde Akinleye & Zoaib Rasool. (2019). Immune checkpoint inhibitors of PD-L1 as cancer therapeutics. Journal of Hematology & Oncology volume 12, Article number: 92 (2019) Chew HY, De Lima PO, Gonzalez Cruz JL, Banushi B, et al. (2020). Endocytosis Inhibition in Humans to Improve Responses to ADCC-Mediating Antibodies. Cell. 2020 Mar 5;180(5):895-914 Elbers JBW, Al-Mamgani A, Tesseslaar MET, van den Brekel MWM, Lange CAH, van der Wal JE, Verheij M, Zuur CL, de Boer JP. (2020). Immuno-radiotherapy with cetuximab and avelumab for advanced stage head and neck squamous cell carcinoma: Results from a phase-I trial. Radiother Oncol. 2020 Jan;142:79-84. Tafuri A, Smith DD, Cacciamani GE, Cole S, Shakir A, Sadeghi S, Vogelzang NJ, Quinn D, Gill PS, Gill IS. (2020). Programmed Death 1 and Programmed Death Ligand 1 Inhibitors in Advanced and Recurrent Urothelial Carcinoma: Meta-analysis of Single-Agent Studies. Clin Genitourin Cancer. 2020 Jan 31. Wang X, Wu S, Chen Y, Shao E, Zhuang T, Lu L, Chen X. (2020). Fatal Adverse Events Associated With Programmed Cell Death Ligand 1 Inhibitors: A Systematic Review and Meta-Analysis. Front Pharmacol. 2020 Jan 31;11:5. Isotype Control: