Pembrolizumab Biosimilar - Research Grade
Bulk Pembrolizumab Biosimilar - Research Grade
Product Benefits:ichorbio's Pembrolizumab Biosimilar - Research Grade is manufactured in a cGMP compliant facility. Click here to view ichorbio’s complete list of anti-PD-1 antibodies and biosimilars.
Size:ichorbio's research grade pembrolizumab biosimilar is available in the following sizes: 5mg, 10mg, 20mg, 50mg, 100mg ichorbio regularly manufactures bulk multi-gram amounts of our pembrolizumab biosimilar - please contact us for pricing.
Target:PD-1
Clone:n/a
Isotype:IgG4 (S228P) kappa
Other Names:Programmed cell death protein 1, Pdcd1, CD279
Host:Humanized
Species Reactivity:Human
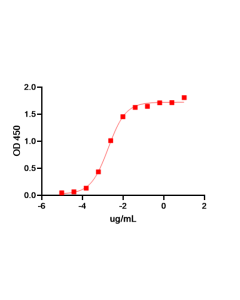
Specificity:Detects human PD-1.
Purification Method:This monoclonal antibody was purified using multi-step affinity chromatography methods such as Protein A or G depending on the species and isotype.
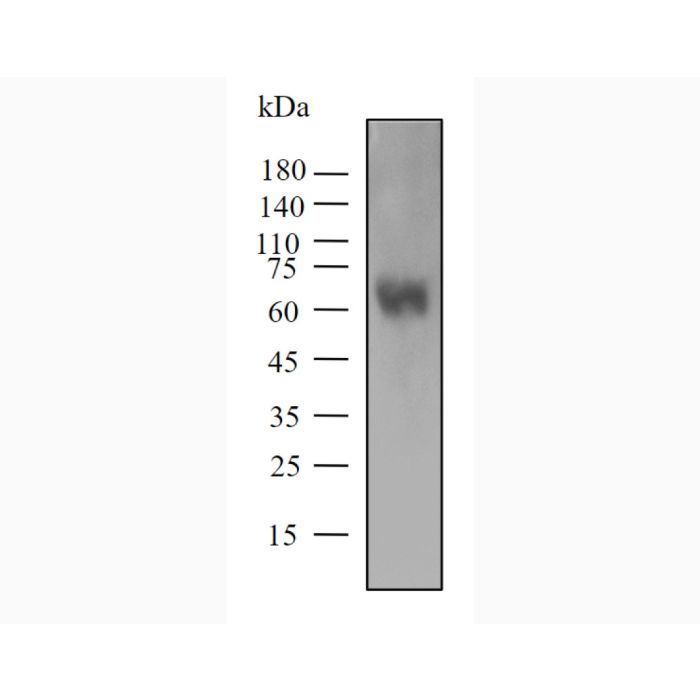
Background:Pembrolizumab biosimilar is a humanized monoclonal antibody that blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2. It is an IgG4 kappa immunoglobulin with an approximate molecular weight of 149 kDa.
Concentration:1.0 - 5.0 mg/ml
Formulation:Sterile, preservative-free, clear to milky white colorless to slightly yellow solution, containing L-histidine , polysorbate 80 and sucrose. pH set to 5.5. BSA and Azide free.
Purity:>95% by SDS-PAGE and HPLC
Endotoxin:≤ 0.75 EU/mg as determined by the LAL method
Storage:This antibody is stable for at least 4 weeks when stored at 2-8°C. For long term storage, aliquot in working volumes without diluting and store at – 20°C or -80°C. Avoid repeated freeze thaw cycles.
Applications:Functional Assays
Application Notes:Each investigator should determine their own optimal working dilution for specific applications.
Isotype Control: Bulk Human IgG4 Isotype Control S228P (IB4) [ICH2257S228P] Use: ichorbio's pembrolizumab biosimilar is for research use only (RUO). Recent Research on Pembrolizumab: Pembrolizumab is a humanized monoclonal IgG4 kappa antibody directed against the human cell surface receptor PD1 (programmed cell death-1). PD1, a transmembrane protein expressed at the surface of effector T-cells, plays a key role in the regulation of immune checkpoints and tumor progression (Kwok et al., 2016). Research surrounding PD1 and its ligands lead to the approval of pembrolizumab for medical use in 2014 for its ability to block PD1 and positively regulate T-cell-mediated immune response against cancer cells.In NSCLC (non-small cell lung cancer), recent research by Lin et al. (2020) investigated the efficacy of the combination therapy of pembrolizumab and NK cell infusion in patients with previously treated advanced cancer. Data showed that the combination treatment significantly improved patients’ overall survival and progression-free survival compared to pembrolizumab alone. The authors also found a higher expression level of PD1 in the peripheral NK and T cells of cancer patients compared to healthy donors. Pembrolizumab in these patients reversed high PD1 expression, increased secretion of IFN-gamma and enhanced immune function. Interestingly, combination therapy also significantly lowered the number of CTCs (circulating tumor cell) and tumor markers.In cervical cancer, two research articles in 2018 showed the presence of PD1-positive T cells in the tumor stroma and high level of PD-L1, its ligand, on the surface or cervical tumor cells (Meng et al., 2018, Feng et al., 2018). Several immunotherapeutic targets are currently under investigation in clinical trials and pembrolizumab was recently approved for the treatment of patients with recurrent and metastatic cervical cancer. In 2019, the phase II basket study Keynote-158 confirmed the antitumor activity of pembrolizumab monotherapy for patients with advanced PD-L1 positive cervical cancer (Chung et al., 2019).In hepatocellular carcinoma, immune checkpoints inhibition is also an area in rapid development especially for patients who do not respond to first-line treatment. In 2018, pembrolizumab was evaluated in patients with advanced HCC, with intolerance to the systemic therapy, sorafenib. In this phase II trial, improvement in overall survival and durable objective response was associated with the presence of PD-L1 in a subset of patients (Hu et al., 2018). These promising results led to the approval of pembrolizumab as a second-line agent for the treatment of advanced HCC. In 2020, the keynote-240 trial confirmed the results obtained previously and despite the lack of statistical significance in overall survival and progression-free survival, it supported a favorable risk-to-benefit ratio in the population tested.In breast cancer, recent research highlighted the importance of immune checkpoints in patient’s response to therapy and long-term survival especially for patients with invasive breast cancer. Pembrolizumab is not yet approved to treat breast cancer patients however it has been tested in several clinical trials for efficacy and safety. For example, in 2019, research by Vinayak et al. investigated the efficacy of nirapirib combined with pembrolizumab. They observed promising antitumor activity results in patients with advanced or metastatic TNBC (triple-negative breast cancer). Similarly, in 2020, Ho et al. tested the impact of pembrolizumab and radiotherapy which was found to be safe and encouraging in patients with poor-prognosis metastatic TNBC.These data indicate the promising opportunities to improve existing immunotherapy for the treatment of patients with difficult to treat cancer even though important research is still needed to improve the selection of patients who may benefit from these treatments. Currently, pembrolizumab is approved to treat 15 types of cancer, including cervical cancer, hepatocellular carcinoma, melanoma, and NSCLC.KEYWORDS: PD1; PDL1; immune checkpoints; non-small cell lung cancer; HCC; cervical cancer; breast cancer; research pembrolizumab; advanced tumors.References: Chung HC, Ros W, Delord JP, Perets R, Italiano A, Shapira-Frommer R, Manzuk L, Piha-Paul SA, Xu L, Zeigenfuss S, Pruitt SK, Leary A. (2019) Efficacy and Safety of Pembrolizumab in Previously Treated Advanced Cervical Cancer: Results From the Phase II KEYNOTE-158 Study. J Clin Oncol. 2019 Jun 10;37(17):1470-1478Emen LA. (2018). Breast Cancer Immunotherapy: Facts and Hopes. Clin Cancer Res. 2018 Feb 1;24(3):511-520Feng, YC, Ji, WL, Yue, N, Huang, YC, and Ma, XM. (2018). The relationship between the PD-1/PD-L1 pathway and DNA mismatch repair in cervical cancer and its clinical significance. Cancer Manag. Res. 10, 105–113.Gerry Kwok, Thomas C. C. Yau, Joanne W. Chiu, Eric Tse & Yok-Lam Kwong. (2016) Pembrolizumab (Keytruda). Hum Vaccin Immunother. 2016 Nov;12(11):2777-2789.Ho AY, Barker CA, Arnold BB, Powell SN, Hu ZI, Gucalp A, Lebron-Zapata L, Wen HY, Kallman C, D'Agnolo A, Zhang Z, Flynn J, Dunn SA, McArthur HL. A phase 2 clinical trial assessing the efficacy and safety of pembrolizumab and radiotherapy in patients with metastatic triple-negative breast cancer. Cancer, 2020 Feb 15;126(4):850-860.Hu AX, Finn RS, Edeline J, Cattan S, Ogasawara S, Palmer D. (2018) KEYNOTE-224 investigators. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol. 2018 Jul;19(7):940–52.Mao Lin, Haihua Luo, Shuzhen Liang, Jibing Chen, Aihua Liu, Lizhi Niu, and Yong Jiang. (2020). Pembrolizumab plus allogeneic NK cells in advanced non-small cell lung cancer patients. J Clin Invest. 2020 Feb 6Meng, Y, Liang, H, Hu, J, Liu, S, Hao, X, Wong, MSK (2018). PD-L1 Expression correlates with tumor infiltrating lymphocytes and response to neoadjuvant chemotherapy in cervical cancer. J. Cancer 9, 2938–2945.Vinayak S, Tolaney SM, Schwartzberg L, Mita M, McCann G, Tan AR7, Wahner-Hendrickson AE, Forero A, Anders C, Wulf GM, Dillon P, Lynce F, Zarwan C, Erban JK, Zhou Y, Buerstatte N, Graham JR, Arora S, Dezube BJ, Telli ML. Open-Label Clinical Trial of Niraparib Combined With Pembrolizumab for Treatment of Advanced or Metastatic Triple-Negative Breast Cancer. JAMA Oncol. 2019 Jun 13.